Brightly colored nests of rings and flares – chemical footprints left by a vitamin solution as it inches its way across a piece of filter paper – are providing biochemists with evidence that synthetic vitamin C is not an adequate substitute for the real thing. The patterns are being produced by a variety of chromatography, a technique long used to separate the components of complex chemicals. A filter paper wick is dipped into a vial of vitamins liquefied with a special solvent. The wick transports the solution to the center of a filter paper disk, where it spreads with the eagerness of water invading a blotter, leaving colored ripples frozen in its wake.
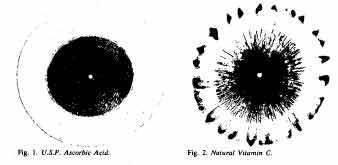
Recently Dr. Justa Smith, a nun and biochemist at Rosary Hill College in Buffalo, has been using the technique on natural and synthetic vitamin C. While the natural vitamin leaves a brightly colored medallion of fluted circles overlaid with flare-like tracings, synthetic vitamin C leaves only drably tinted, unadorned rings.
The complexity of its patterns suggests that the natural vitamin contains something the artificial vitamin does not, but no one is quite sure what. Dr. Smith suspected that the pattern might indicate the presence of a protein. Indeed, on testing, she found there was a protein in the natural vitamin. (The synthetic counterpart has no proteins whatsoever.) What’s more, the protein showed signs of being either wholly or partially an enzyme – one of those critical instigators of the chemical matings and separations essential for life.
But the protein in natural vitamin C may not completely explain the vitamin’s intricate trail. Dr. Smith obtained equally elaborate patterns from clay – a substance without proteins or organic components. She imagines that clay’s mineral-richness may be responsible for its chromatographic complexity, but that is only a guess.
The nature of natural vitamin C’s extra components may remain enigmatic, but there is nothing enigmatic about its importance to health. Dr. Szent-Gyorgyi, a highly respected biochemist who directed the research facility at Woods Hole, fed one group of rats natural vitamin C, while he fed a second group the synthetic substitute. The rats fed the artificial vitamin showed a tendency to develop bruises. The rats fed on natural C, however, showed no tendency to bruise.
Here are more points to keep in mind if you’d like to cash in on natural vitamin C’s mystery element. BHA and BHT, two popular preservatives so potent that only the packaging material need contain them, both destroy the complexities of the natural vitamin’s chromatographic track. Aging the vitamin C also causes its chemical complexities to disappear. So try to get your vitamin C from fresh, natural sources, untouched by preservatives, or you may end up missing a valuable nutritional bonus.
(Dr. Linus Pauling, who crusaded evangelically for vitamin C, argued that massive doses bring a wide rang of beneficial effects, but he claimed there is no difference between the natural and synthetic vitamin. “If anyone could get me eyeball to eyeball meeting with Dr. Pauling,” says Dr. Smith, “I’d sure appreciate it.”





